Vapour produced above the surface of a boiling cargo due to evaporation is called Boil off and is caused by heat ingress or a drop in pressure.
Heat is transferred to the cargo through the insulation.
Cargo moving or sloshing in the tank.
Vapour extracted from a tank reduces pressure
Reduced vapour pressure encourages more vapour production.
If vapour production were allowed to continue then the safety valves would lift on the tank.
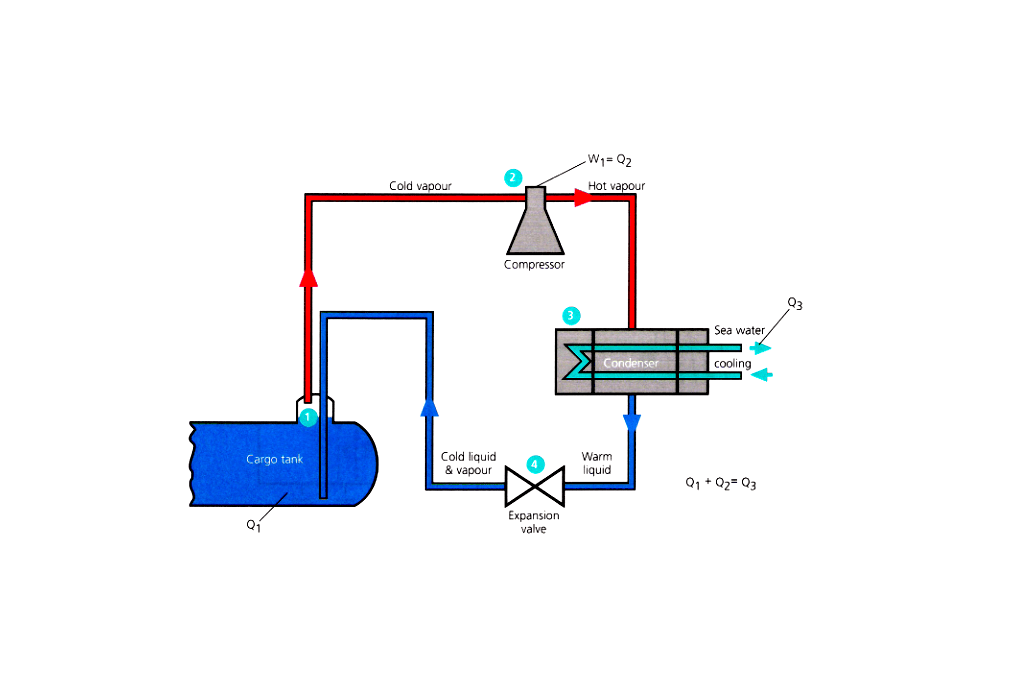
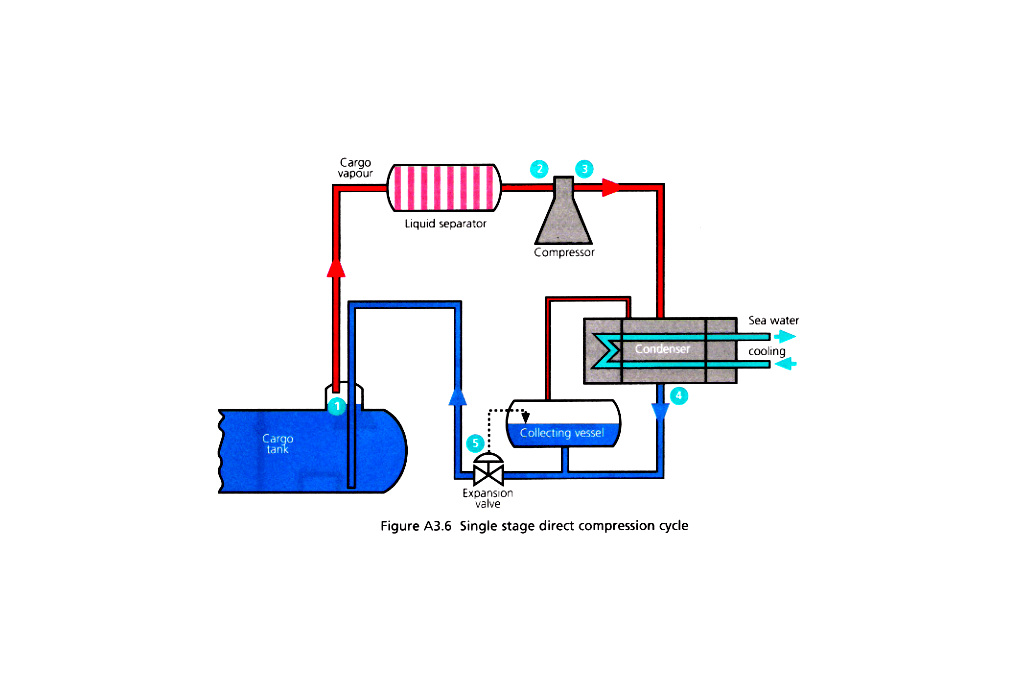
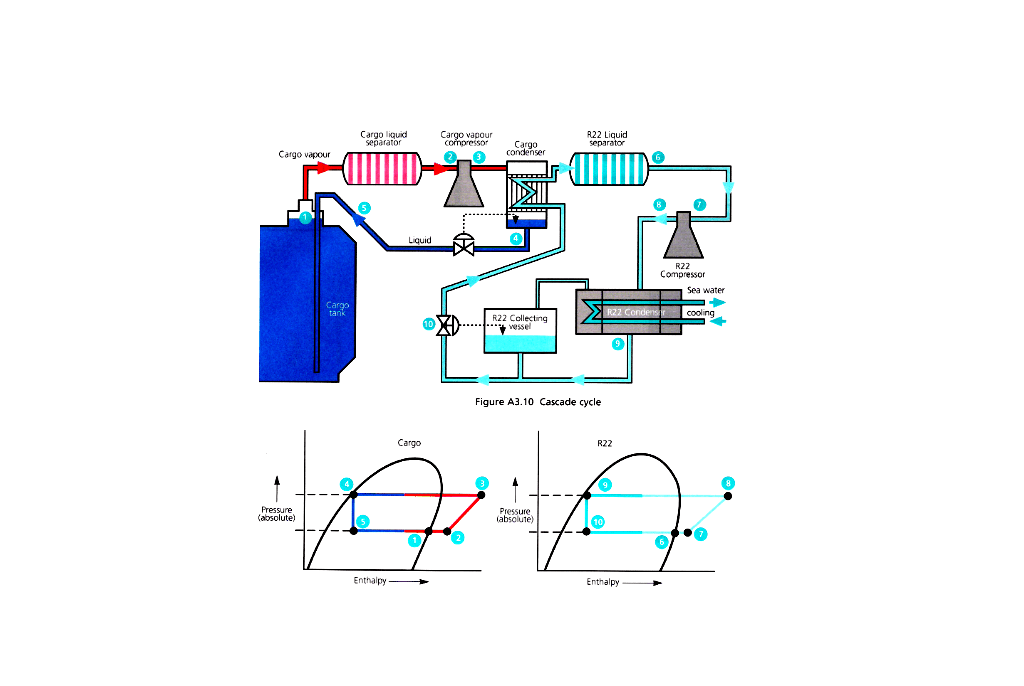
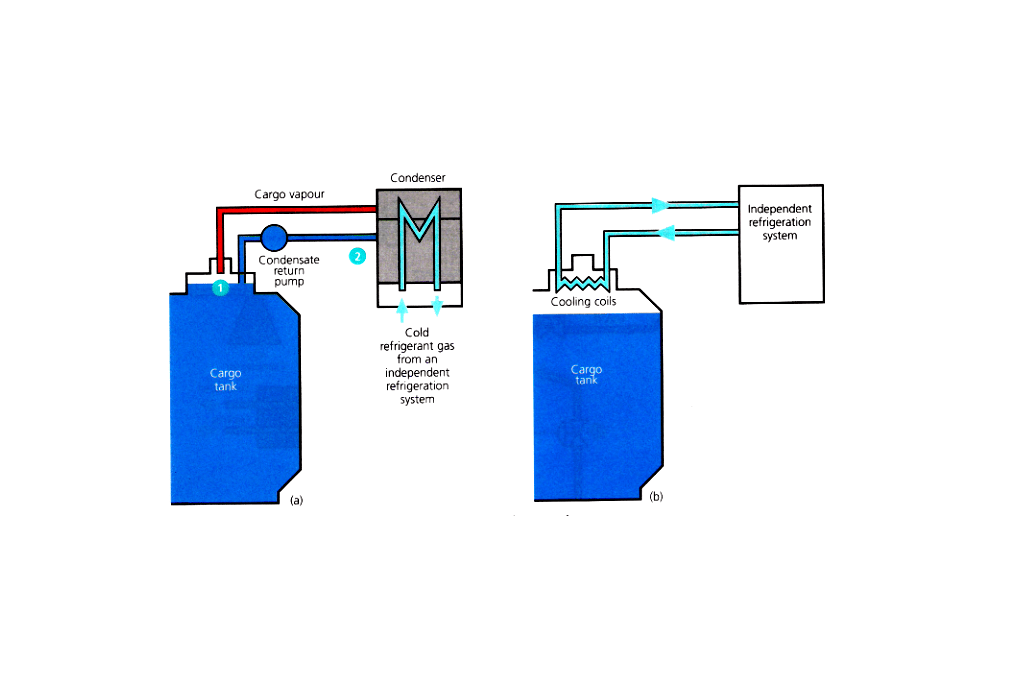
Reliquefication is carried out on board for the following operational reasons:
- If the installation does not have the ability to handle vapour produced whilst loading the liquid into the tanks.
- Whilst at sea, to prevent the pressure rising in the tanks due to the cargo taking in heat through the tank insulation, then the vapour can be condensed and returned to the tank.
- If the discharge port requires the cargo to be at a lower than carriage temperature for operational reasons.
- To maintain the cargo tanks at a low temperature on the ballast voyage, or, cool them for loading.
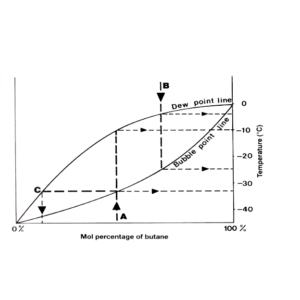
Bubble Point
Temperature at which the liquid will begin to boil as the temperature rises (at a given pressure)
Dew Point
Temperature at which a vapour begins to condense as the temperature decreases (at a given pressure)
If a mixture of 75% butane is heated it boils at -15ºC. Vapour in equilibrium at this temperature has a composition of about 50% butane. If this vapour is condensed, it will give a liquid that will boil at about -27ºC. Vapour equilibrium at this temperature has a composition of about 25% butane.
Propane – Ethane
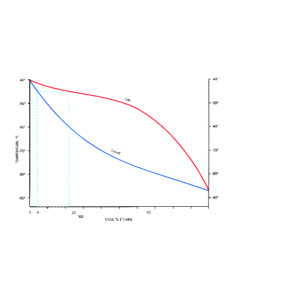
Equilibrium
Mole % propane =
100 – Mole % ethane
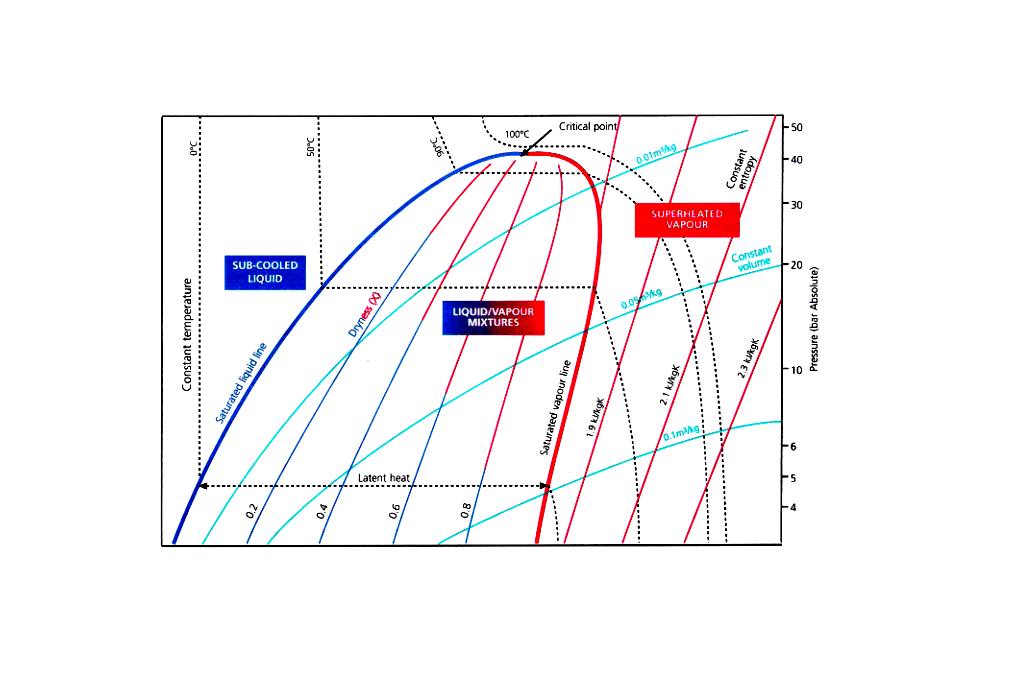
Enthalpy
Enthalpy of a substance is a measure of its thermodynamic heat (or energy) content, whether the substance is liquid or vapour or a combination of the two.
Duration
Cargo temperature on loading
Required cargo temperature on discharge
Cargo composition
Weather conditions
Mollier Diagram Exercise – Ammonia (Simple Cycle)
Point 1
Temp -33°C, Press 1.031 bar, Enthalpy 1636 kJ/kg, Vapour Den 0.905 kg/ m3
Point 2
Temp 30°C, Press 11.7 bars, Enthalpy 1999kJ/kg
Point 3
Temp 30°C, Press 11.7 bars, Enthalpy 560 kJ/kg
Point 4
Temp -33°C, Press 1.031 bar, Enthalpy 560 kJ/kg, Dryness fraction 22%
Diff in Enthalpy = 1076 kJ/kg (Vapour density = 0.880 kg/m3, heat per unit volume = 974 kJ/kg)
Energy absorbed in compression = 363 kJ/kg
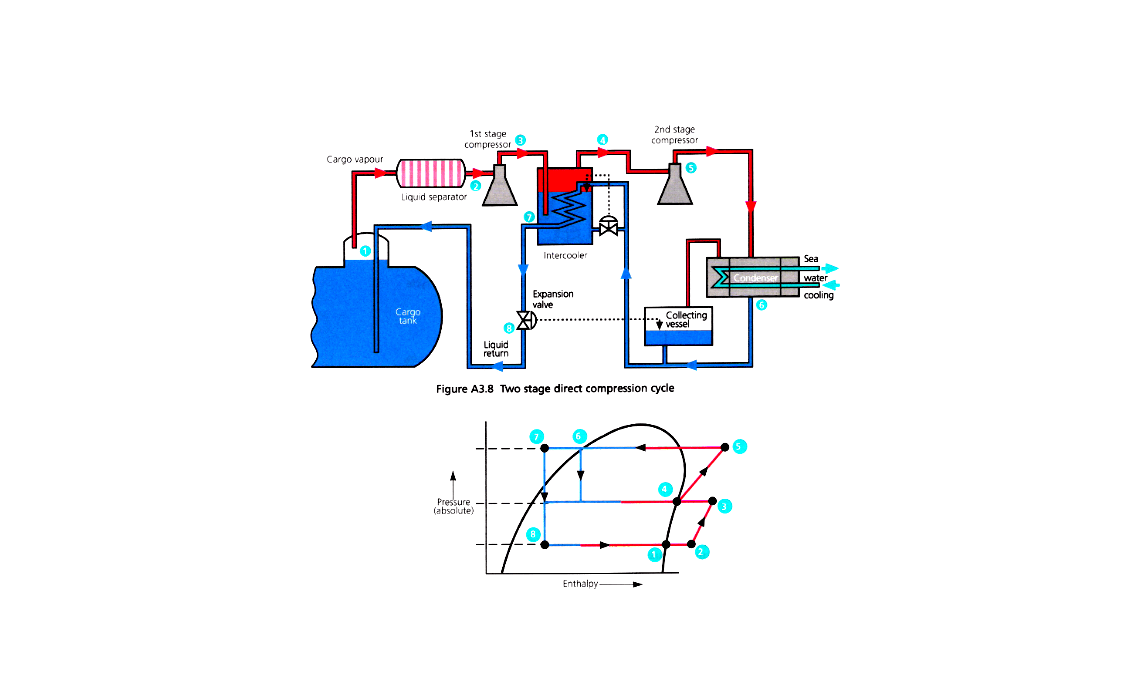
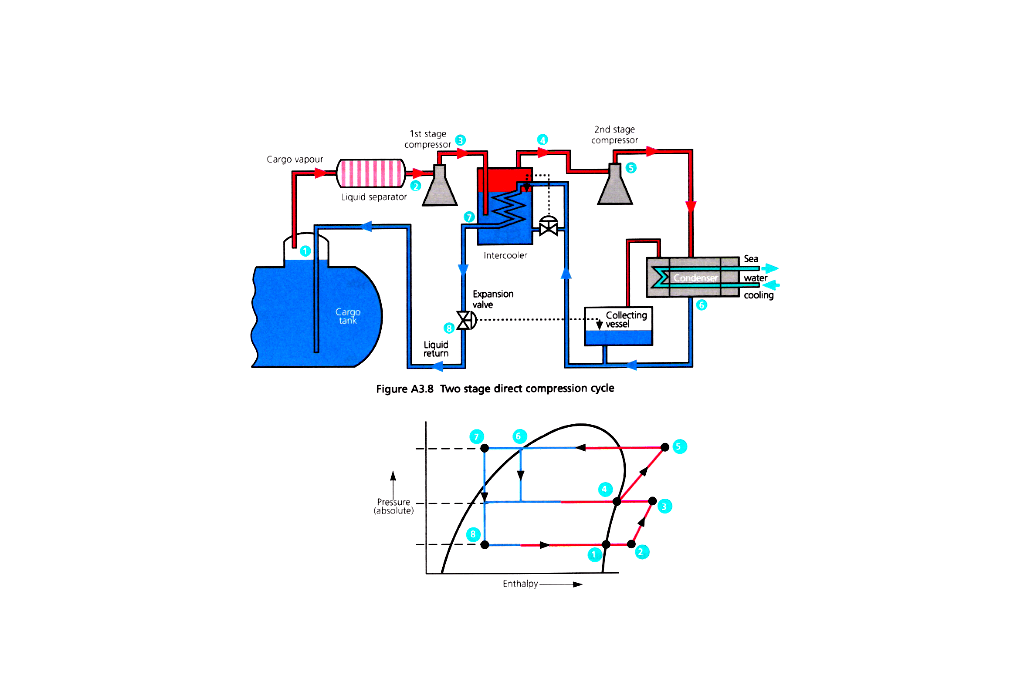
Two Stage
1 – Collect vapour (Boil off)
2 – Compress 1st stage
3 – Superheated
4 – Intercooler (reduce suction pressure of 2nd stage)
5 – Compress 2nd stage (further superheated)
6 – Seawater cooling and condensing
7 – Condensed liquid can be used as intercooler coolant
8 – Ambient temperature liquid collected and passed through expansion valve
LNG
Used for propulsion. Not reliquefied economically. Boil off used for propulsion
Incondensible gases
Oxygen, Nitrogen and low boiling point cargo components (e.g. ethane)